Started on January 1, 2017, the SPIDIA4P project builds on SPIDIA’s results and is funded by the European Union’s Horizon 2020 research and innovation programme. The consortium of 19 highly experienced partners from private industry including SMEs, public institutions and one European Standards Organisation is again coordinated by QIAGEN GmbH. It plans to initiate, develop and implement a comprehensive portfolio of an additional 14 pan-European pre-analytical CEN/TS and ISO/IS documents as well as External Quality Assessment schemes (EQAs), addressing the important pre-analytical workflows applied to personalised medicine.
SPIDIA4P is coordinated by QIAGEN GmbH, Germany, a diagnostic and life science company, with extensive know how in pre-analytical and analytical procedures to gain molecular insights from clinical and biological samples. Sub-contractor ACIES supports the coordinator for effective project management and has long standing experience in research management, project and finance controlling as well as meeting organisation.
The SPIDIA4P consortium will closely coordinate with large European public research consortia to obtain access to research and validation studies data, serving as evidence for the new standards developments for achieving improvements of diagnosis, patient stratification and prognosis of disease outcome.
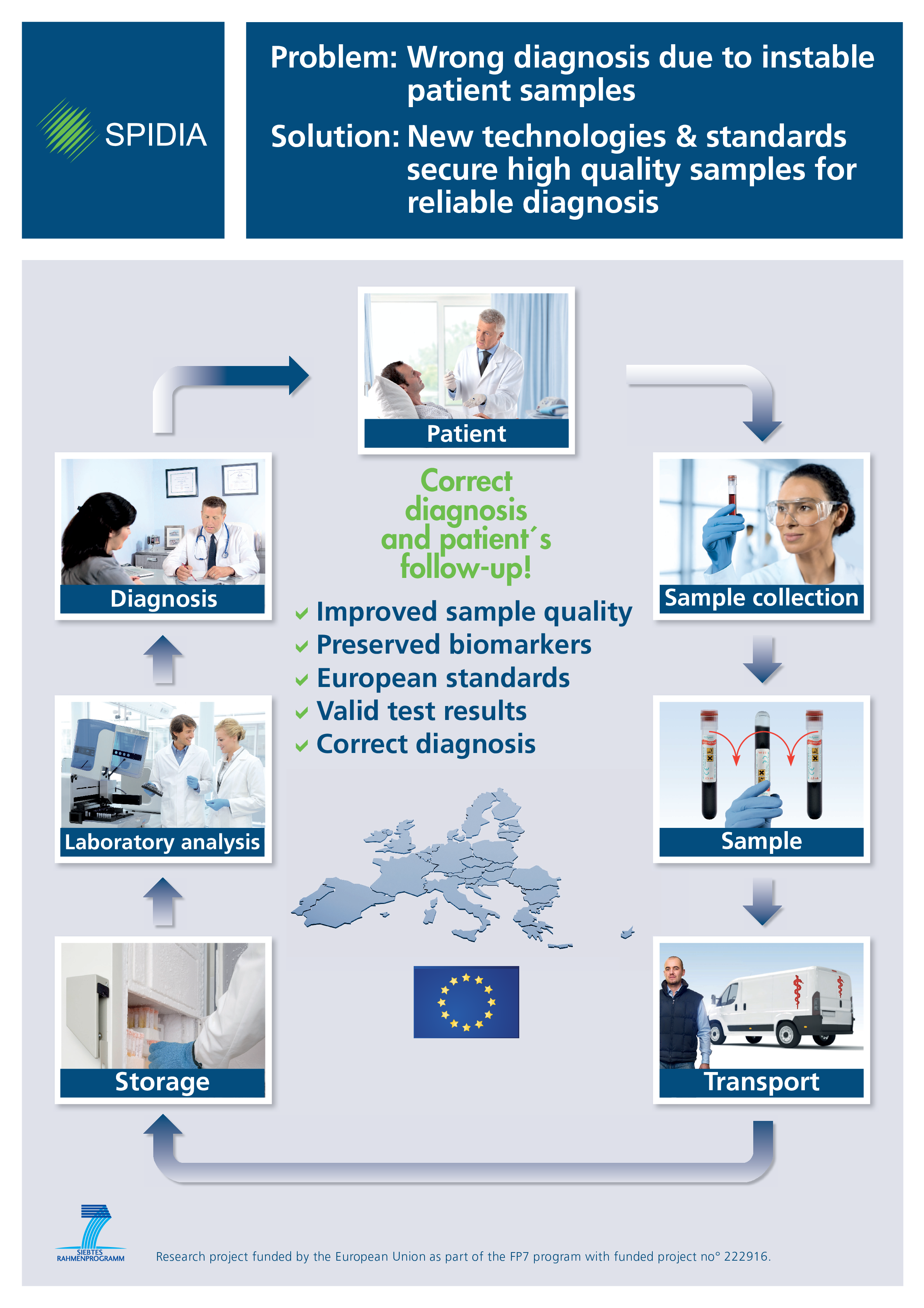
OUR GOAL: IMPROVEMENT OF THE GLOBAL HEALTHCARE SYSTEMS by development of selected high priority pre-analytical CEN and ISO standard documents as well as corresponding External Quality Assessment (EQA) schemes and implementation tools.